
Mycotoxins are toxic substances produced by fungi (molds) growing on grain, feed, or food in the field or in storage. Mycotoxins may be detrimental to the health of both animals and humans. Although thousands of molds are capable of growing on stored grain, only a few mold species produce mycotoxins. There are many different mycotoxins, but most are rare in Midwest grain. In Indiana, the mycotoxins of concern are: aflatoxin, deoxynivalenol (DON or sometimes called vomitoxin), zearalenone, and fumonisin. All four of these mycotoxins are associated with ear rot diseases.
Fields affected with significant amounts of ear rot should be harvested and handled separately. Grain should be harvested as early as possible and dried to 15% moisture or lower. Usually, specific environmental conditions are required for mycotoxin production in pre-harvest grain. Being aware of these conditions and the amount of disease in the preharvest crop should help in judging the likelihood of a mycotoxin problem. Mycotoxins can be produced in storage; however, this situation is almost always associated with improper drying and/or storage conditions.
Aflatoxin is a potent liver toxin and known to cause cancer in animals. In swine, aflatoxin can cause reduced weight gain, reduced ability to resist diseases, hepatitis. and death. The Food and Drug Administration (FDA) has established action levels of 20 parts per billion (ppb) for grain and feed products, and 0.5 ppb for milk. Grain, feed, or milk containing aflatoxin at or above these levels cannot be sold for food or feed in interstate sales. Mixing aflatoxin contaminated grain with sound grain for sale is illegal. Corn and other grain with less than 20 ppb aflatoxin can be sold as normal grain. Recommended limits in feed are: 20 ppb for dairy animals; 100 ppb for breeding cattle, breeding swine, and mature poultry; and 300 ppb for finishing cattle and swine.
Aflatoxins are produced by the fungus Aspergillus flavus. This fungus causes a disease in preharvest corn known as Aspergillus ear rot. Aspergillus ear rot and aflatoxin are a concern mainly in drought years and are more commonly found in the southeastern and southwestern portions of the United States. Although Aspergillus flavus is a common fungus growing as a saprophyte on dead plant debris, infection and aflatoxin production in preharvest corn occur almost exclusively in years when plants are severely stressed by drought. Aflatoxin production in the field is favored by high grain moisture, temperatures in the range of 80-100 F, severe drought stress, nitrogen deficiency, and significant insect damage. Except in hot, dry years, aflatoxin in the Midwest is almost exclusively associated with improper storage of grain or feed. Aflatoxin production in stored grain should not occur if grain is sufficiently dried to 13-14% moisture and maintained at that level.

Many times, grain is initially screened for aflatoxin by using a black light to detect kernels or portions of kernels that glow with a bright green yellow fluorescence (BGYF). This is strictly a presumptive test and indicates only that the causal fungus, Aspergillus flavus, was growing on the living kernel and does not indicate the presence of aflatoxin or other mycotoxins. BGYF is best observed in cracked corn rather than whole kernels. When examining corn for BGYF, one should have a color standard or an authentic BGYF for comparison. The causal fungus occasionally may be observed growing on kernels, but its presence does not guarantee that aflatoxin is present. Therefore, a chemical test is necessary for the actual detection of aflatoxin.
Deoxynivalenol is also known as vomitoxin or DON. This mycotoxin causes reduced animal feeding and weight gain (especially swine) at levels as low as 1-3 parts per million (ppm). Vomiting and total feed refusal does not occur until DON concentrations are much higher (> 10 ppm). The FDA has recommended that DON levels not exceed 1 ppm for human food. The following limits have been recommended:

Deoxynivalenol is produced by the fungus Gibberella zeae, also known as Fusarium graminearum. This fungus causes Gibberella ear rot on corn and head scab on wheat. Gibberella ear rot is enhanced by cool, wet weather during the period just after silking. The disease is often more severe in fields where corn follows corn, or corn follows wheat, especially if the previous crop was affected with Gibberella ear rot, stalk rot, or head scab, and the infected residues were left on the soil surface.
Prior to harvest, Gibberella ear rot is readily identified by examining the intact ears. The disease is visible as a pink or reddish mold at the tip of the ears when the husk is peeled back. If the disease is severe, the silk will adhere to the ear and may be reddish in color. Once the grain is shelled, it is difficult to determine by visual examination if the grain is infected.
After harvest, production of DON is favored by grain moisture of 20.5% or more, and temperatures of 70-85 F. Grain containing DON also may contain smaller amounts of the mycotoxin zearalenone. Once the grain is harvested, growth of the fungus that produces DON is effectively halted if the grain is dried to 15% moisture or lower.
Zearalenone is an estrogenic mycotoxin, and can cause infertility, abortion or other breeding problems when fed to swine. As little as 0.1 to 5 ppm zearalenone in a feed ration may produce estrogenic syndrome in swine. Also, uterine prolapse can occur in young pigs with concentrations as low as 1 ppm zearalenone. There have been no limits or recommended levels placed on grain used for human consumption.
Zearalenone can be produced in preharvest corn or wheat by strains of Gibberella zeae (Fusarium graminearum), however it is usually produced in low concentrations. Often, both DON and zearalenone occur together. In stored grain, production is favored by grain moisture in excess of 20.5% and 70-85 F temperatures. As with DON, the growth of the fungus that produces zearalenone is effectively halted if the grain is dried to 15% moisture or lower.
Fumonisin is a recently discovered mycotoxin that may be the cause of equine leukoencephalomalacia (blind staggers) in horses, donkeys and mules, and pulmonary edema in swine. There is also evidence linking fumonisin to cancer in humans. No restrictions are currently placed on food because little is known about the effects of fumonisin on humans. Fumonisin levels in feed are recommended to be no higher than 5 ppm for horses, 10 ppm for swine, and 50 ppm for cattle.
The fungus that produces fumonisin, Fusarium moniliforme, causes Fusarium ear rot of corn, the most common and widespread ear rot in the Midwest: affecting as much as 90% of the corn fields. Diseased ears will have signs of the fungus growing on and between individual kernels or groups of kernels. The fungus will appear cottony white or light grey. Diseased kernels will become grey to brown.

The conditions under which fumonisin is produced are not known. Fusarium moniliforme can grow over a wide range of temperatures and moistures, and some strains of the fungus are more efficient producers of fumonisin than others. The severity of Fusarium ear rot is greatest in dry years. Kernels that are infected with Fusarium moniliforme are more brittle than healthy kernels. As a result, these infected kernels break during harvest and drying, and end up in the screenings. For this reason horses and swine should not be fed corn screenings.
Unlike the other mycotoxins associated with corn, high fumonisin concentrations have also been found in seemingly healthy corn kernels. Examination of such corn reveals that Fusarium moniliforme is present in these kernels, even though there are no visible symptoms or signs of disease. Because of this risk, horse owners would be advised to check for fumonisin using one of the commercial test kits.
The first step in mycotoxin determination is the sampling of the grain. Particular attention should be given to the sampling procedure because sampling error will be the greatest source of variance in the analytical procedure. This variance is primarily due to the uneven distribution of the mycotoxin contaminated kernels within a lot of grain or feed. The ideal sampling procedure should assure the highest probability of detecting mycotoxins even when contamination is low.
One method of sampling grain is to use a probe sampler. Since mold growth usually occurs in spots in the grain lot, best sampling is done on recently blended lots of grain. Another method of sampling is to collect small samples from the stream of grain as it is moved in or out of the bins.
With both sampling methods, the collected grain is pooled into a large aggregate sample that represents the lot. For shelled corn, it is recommended that the aggregate sample be about 10 pounds. The aggregate sample should be coarsely ground. Most analytical procedures need only about 25 grams (0.9 ounces) of ground corn, so it is important that the aggregate sample be thoroughly mixed after grinding. A one or two pound subsample is then taken and it is ground to the texture of instant coffee. From this subsample, a final sample is taken for analysis.
The most accurate and reliable method of analyzing mycotoxin levels is to send test samples to professional laboratories for analysis. This. however, is often not practical because of the cost or the time required for analysis. Because of the need for fast determination of mycotoxin levels, a variety of mycotoxin test kits are sold that are easy to use and relatively inexpensive.
Before purchasing a mycotoxin test kit, it is important that one determine what type of information is required. Even though the technology in the analysis of mycotoxin kits is constantly improving, the information derived from the kits is basically of two types; quantification or threshold levels.
A test kit that determines the exact amount of mycotoxin is needed by industrial grain handlers who must document the quality of their starting materials or finished product. Special equipment must be purchased that measures the mycotoxins levels.
In contrast, livestock handlers often need to know only that the level of mycotoxin in their feed is below the threshold levels for their animals. This information can be obtained from kits that provide a quick yes or no answer at a specific threshold level of mycotoxin without the use of special equipment.
Mycotoxin test kits work by one of two methods: thin layer chromatography (TLC) or immunochemistry. While the TLC method has been around for a long time, it has both advantages and disadvantages compared to the immunochemical methods. One advantage of the TLC method is that more than one mycotoxin can be tested for with a single kit. With the immunochemical kits, a separate kit must be purchased for each mycotoxin.
A second advantage of the TLC method is that the mycotoxin is actually visualized. The immunochemical kits detect the mycotoxin indirectly and thus are more prone to error. Error can be reduced by purchasing test kits from reputable companies and carefully following the instructions.
The advantages of the immunochemical methods are their ease of use and the short time required for the analysis. For those who only wish to know that the toxin concentration in a sample is above or below the toxic levels recommended in animal feed, the immunochemical test kits are probably the method of choice.
The following is a summary of mycotoxin kits from three commercial sources. These detection kits illustrate the different technologies being used. One should realize that these technologies are constantly being improved and that these companies are striving to make the detection kits more accurate, sensitive, easier to use, less expensive, and more rapid.
Reference to products in this publication is not intended to be an endorsement to the exclusion of others which may be similar. Persons using such products assume responsibility for their use in accordance with current label directions of the manufacturer.
Romer Labs Inc. 1301 Stylemaster Drive Union, Missouri 63084 (314) 583-8600
Test kits are available for aflatoxin, vomitoxin (DON), and zearalenone. Price: $379 for 25 tests; all three mycotoxins may be detected with one kit.
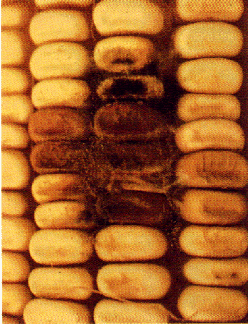
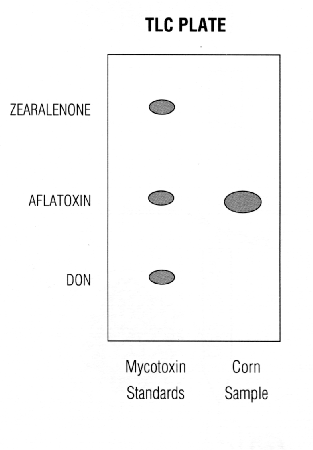
Romer's "MYCOTEST" uses TLC technology. Corn samples are ground and extracted. The extracts are then spotted onto a TLC plate. One TLC plate can be spotted with extracts from several corn samples. The TLC plate is developed, dipped into an aluminum chloride solution and heated. The mycotoxins are then visualized by viewing the plate under long-wave ultraviolet irradiation (black light). Mycotoxin standards are also available making it possible to visually estimate the quantity of the mycotoxins present. The corn sample illustrated below contains aflatoxin at approximately three times the standard.
Romer also sells a test kit (AFLA-CUP) for aflatoxin that uses the ELISA technology (see discussion of the Neogen products).
Neogen Corporation 620 Lesher Place Lansing, Michigan 48912-1509 (800) 234-5333
Test kits are available for aflatoxin, vomitoxin (DON), zearalenone. fumonisin, T-2 toxin and ochratoxin. Price: $80-130 for 24 test wells; each mycotoxin requires a separate kit.
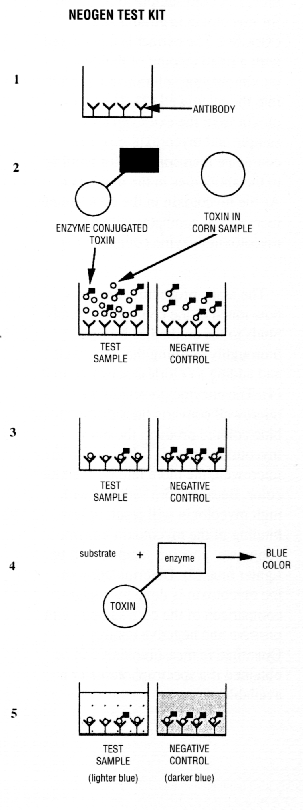
Neogen's "AGRI-SCREEN" and "VERATOX" use ELISA technology. Antibodies specific for a mycotoxin are adhered to the wall of a microwell (1). A solution of mycotoxin, chemically conjugated to an enzyme. is provided with the kit (2). A corn sample to be tested for mycotoxin is ground and extracted. The extract is then mixed with a fixed amount of the mycotoxin-enzyme solution and placed into the microwell (2). The mycotoxin from the extracted corn sample and mycotoxin-enzyme conjugate then compete for binding to the antibodies in the microwell. As the mycotoxin in the corn sample increases, it competes with the mycotoxin-enzyme conjugate (3).
The assay procedure measures how much of the conjugate actually binds to the antibodies by first thoroughly washing the microwell and adding a colorless substrate to it (4). The enzyme present in the microwell converts the substrate to a blue colored product; the more mycotoxin-enzyme conjugate in the microwell, the more intense the blue color. Because corn samples with high mycotoxin will result in less binding of the mycotoxin-enzyme conjugate, positive samples will be lighter blue (5). Determination of the mycotoxin is done by visual comparison of the corn sample with positive and negative controls. Quantitative measurements can be obtained if a spectrophotometer is available.
Vicam 29 Mystic Avenue Sommerville, Massachusetts 02145 (800) 338-4381
Test kits are available for aflatoxin, zearalenone, ochratoxin, and fumonisin. Price: columns for each test are 57.50-10; each mycotoxin requires a separate test. Other needed materials are sold separately.
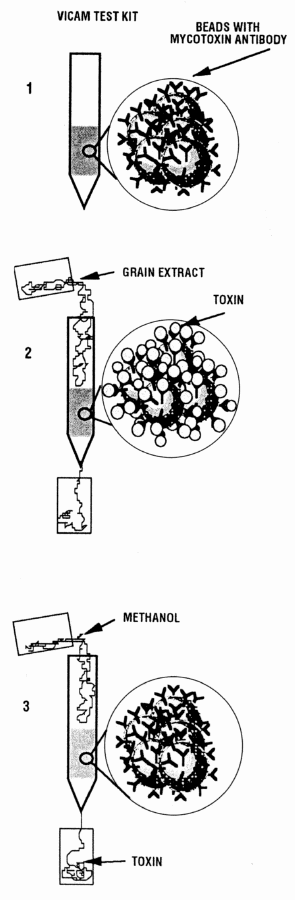
Vicam's columns use immuno-affinity chromatography technology. The columns contain beads chemically fused to antibodies specific for the mycotoxins (1). A corn sample is ground and extracted with a methanol: water solution. The extract is then run through the affinity column and the mycotoxin sticks by binding to the antibody beads (2). Other materials in the extract do not stick and are washed off the column. The mycotoxin is then removed from the column using methanol (3). To visualize and measure the level of mycotoxin, a derivative of the mycotoxin must be made and measured with a fluorometer.
Once mycotoxins are present in grain, there are no practical methods for decontamination. The grain should be stored separate from uncontaminated grain. Getting an accurate measurement of the mycotoxin concentration will help in determining how the grain can be used. If the toxin levels are not excessively high, on-farm blending of the contaminated grain with uncontaminated grain is an option. The contaminated grain also can be fed to animals that are less sensitive to the mycotoxin.
If you have questions about mycotoxins or the use of mycotoxin tests, call your county extension educator or the Plant and Pest Diagnostic Laboratory (P&PDL) at Purdue University.
ID-189 Moldy Corn Poisoning in Horses
(Equine Leukoencephalomalacia) and PIH-129 Mycotoxins and Swine Performance from the Media Distribution Center, (317) 494-6794.
New 7/94
Cooperative Extension Work in Agriculture and Home Economics, State of Indiana, Purdue University and U.S. Department of Agriculture Cooperating. H.A. Wadsworth, Director, West Lafayette, IN. Issued in furtherance of the Acts of May 8 and June 30, 1914. It is the policy of the Cooperative Extension Service of Purdue University that all persons shall have equal opportunity and access to our programs and facilities.