
WQ-19

Pesticides are designed to stop or limit pest occurrence. Insecticides kill insects, while herbicides kill weeds and fungicides suppress or kill fungi. All of these products are pesticides.
Most of us recognize that pesticides allow agricultural producers to improve the quality, quantity and diversity of our food supply. In addition, pesticides are used in the timber, turf, horticulture, aquatic and structural pest control (exterminating) industries to protect the places where we live, work and play. Many homeowners and home gardeners also apply pesticides to their houses, yards and animals.
It is important to understand the fate of a pesticide once it is applied, as this allows applicators to select the most effective and environmentally safe product. This publication provides a general overview of how pesticides interact with the environment.
The goal of pesticide use is to apply products that will remain in the target area long enough to control the specific pest(s) and then degrade into harmless compounds in the soil, air or water without contaminating the environment.
Once applied, many pesticides are mobile in the environment. This movement can be beneficial if the pesticide is carried to a specific target area, like a plant's root zone, or if it helps to ensure that degradation occurs at the proper time and place. Sometimes, however, nontarget insects, plants and other organisms come into contact with the pesticide. This can result in reduced control of the target pest and injury to nontarget plants and animals.
Any remaining pesticide or pesticide residue can move along one, several or all of the pathways described in Figure 1. This movement is a complex process and, combined with several other factors, influences a pesticide's fate. These factors include:
- properties of the pesticide
- various conditions at the application site
- application method
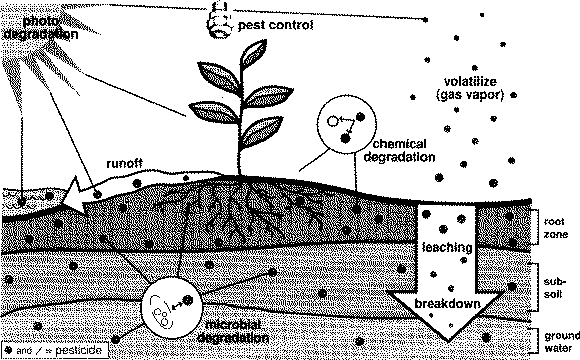
Impairment to the environment may occur when a pesticide overloads a pathway.
Four major pesticide properties important to a pesticide's fate after application are vapor pressure, sorption, water solubility and persistence.
Vapor Pressure (volatility) is the tendency of a pesticide to change to a gas. Pesticides with high vapor pressure are more likely to volatilize and escape into the atmosphere.
Sorption is the attraction of a pesticide to inorganic and organic soil surfaces. When pesticides are sorbed to soils, they are less likely to leach.
Water Solubility expresses the amount of pesticide that will dissolve in a given amount of water. Pesticides with large water solubility generally have lower sorption. This makes them more mobile in the environment and more readily available for leaching to ground water or to running off with surface water.
Persistence refers to the amount of time a pesticide remains in the environment. It is measured by half-life. More persistent pesticides that have longer half-lives can pose a greater threat to the environment since they remain there longer. Persistence is a function of the chemical and biological degradation processes, which break down the pesticide into less harmful compounds (Figure 2).

To maximize pesticide effectiveness and minimize potential environmental concerns, the properties of the pesticide must be compatible with the physical, chemical and biological conditions of the application site. Together, these four factors determine how a pesticide should be applied and influence its ultimate environmental fate.
For pesticides used in crop or plant pest control, the following conditions need to be considered:
- physical and chemical properties of the soil
- geologic layers and surrounding water sources
- environmental conditions
Texture is the percent of sand, silt and clay in soil. The rate that water moves through soil, or its permeability, is affected by texture.
Sandy soils are usually highly permeable because they are coarse textured, or composed mainly of large particles. In general, the sandier the soil, the higher the permeability. Soils high in clay and silt, on the other hand, are fine textured. They are composed mainly of small particles that generally have only tiny gaps for water movement. Soils with high percentages of clay and silt usually have lower permeability (slow flow).
When water containing a pesticide moves through the soil at a slower rate, the potential for pesticide sorption increases. This decreases the threat of contamination from leaching. However, extremely low permeability can increase the chance of pesticide runoff.
Clay soils also can contain more organic matter. Organic matter is the portion of the soil formed from decomposing plant and animal residues, as well as soil microorganisms (bacteria, fungi, etc.) and their by-products.
Although organic matter generally makes up less than five percent of most soils, it improves physical characteristics of the soil such as structure and permeability. Good soil structure and permeability reduce the potential for soil erosion and limit pesticide runoff.
Organic matter is also the most chemically active part of the soil. Its chemical reactivity and large surface area make organic matter an ideal environment for pesticide sorption. Depending on the pesticide and targeted pests, this high sorption capacity can be either beneficial or harmful.
Soil pH (its relative acidity or alkalinity) can have a profound effect on the behavior of some pesticides. Fortunately, most agricultural fields are maintained at a pH level acceptable for pesticide application.
Characteristics of geological layers beneath soils can vary. An understanding of these formations is another key element in selecting the proper pesticide.
The shallower the depth to ground water, the less soil there is to act as a filter. When pesticides are exposed to soil layers with greater depths, more opportunity exists for the chemicals to dilute, degrade or sorb before entering ground water.
Nearby surface water is more susceptible to contamination when pesticides are applied to highly erodible soils or to over-irrigated or rain-soaked fields. Using the wrong pesticide or an incorrect application method in these situations increases the risk for contamination. Managing crop residues and maintaining grass waterways and filter strips help to protect surface waters from sediment and pesticide pollution.
Adverse weather conditions can reduce pesticide performance and have a negative impact on the environmental fate of a pesticide. Applicators need to utilize management methods and pesticide products that account for the climatic conditions at the time of pesticide application.
Heavy or sustained rainfall or irrigation shortly after application can increase the risk of contamination from runoff, leaching and volatilization. This often results in the need for additional applications. It is important to note that many herbicides require a certain amount of water to transport and activate the chemical compounds in a target area.
Extremely high and low soil temperatures adversely affect the degradation rate of some pesticides, as well as interfere with other areas of pesticide performance. During very hot weather, it is important to safeguard against volatilization by using pesticides that have strong sorption and low vapor pressure.
Volatilization can also be a problem in windy weather. Pesticides should not be applied in these conditions. Drift, which results when stray pesticide particles fall onto nontarget species (including humans) can also be a problem. Many pesticides are incorporated, or applied directly into the soil, to decrease the potential for drift and volatilization.
The challenge to all pesticide applicators is to maximize the benefits of pesticides through sound management while reducing environmental risks. Keeping current on the latest pesticide information and understanding the interaction of the physical, chemical and biological factors introduced in this publication provide some of the necessary tools to accomplish this.
Applicators should take all the necessary preventive measures to ensure that pesticides stay on-site. Careful transportation, secure storage, proper disposal of containers and rinsate, and spill prevention are basic elements of safe pesticide use. A careful applicator will also know the location of sensitive areas, including sinkholes, depressions, wells, surface water, public institutions and private buildings. A buffer zone should be in effect when applying pesticides around these sites.
Pesticide applicators who read, understand and follow product labels can minimize risks to human health and the environment. Since the pesticide label is a binding legal document, strictly following label directions from purchase to container disposal also guarantees compliance to state and federal regulations.
In most states, applicators are not able to purchase certain higher risk pesticides unless they have obtained special certificates or licenses that must be renewed at regular intervals. Certification is a means of ensuring that persons who apply these restricted-use pesticides possess the knowledge to do so in a safe, judicious and effective manner. With certain qualifications, private and commercial noncertified applicators may apply restricted-use pesticides or make pesticide applications to the property of another if they do so under the direct supervision of a certified, licensed applicator.
Selecting a pesticide product depends on more than its cost and effectiveness. Since the ultimate environmental fate of pesticides is both site- and product-specific, applicators should account for all the potential risks of pesticide use. This consideration of the environment in the decision-making process shows a commitment toward responsible stewardship that can decrease further contamination of our soil, air and water resources.
For more information, contact your local Extension educator or Purdue Pesticide Programs, (317) 494-1284. The following Purdue University Cooperative Extension publications may also prove helpful.
- WQ 1 "Water Testing Laboratories"
- WQ 2 "What Is Ground Water?"
- WQ 3 "How to Take a Water Sample"
- WQ 4 "Why Test Your Water?"
- WQ 5 "Interpreting Water Test Results Part One: Inorganic Materials"
- WQ 6 "Buying Home Water Equipment"
- WQ 7 "Animal Agriculture's Effect on Water Quality Pastures and Feedlots"
- WQ 8 "Animal Agriculture's Effect on Water Quality Waste Storage"
- WQ 9 "Water Quality for Animals"
- WQ 10 "Wetlands and Water Quality"
- WQ 11 "Sulphur Water Control"
- WQ 12 "Distillation For Home Water Treatment"
- WQ 13 "Home Water Treatment Using Activated Carbon"
- WQ 14 "Reverse Osmosis for Home Treatment of Drinking Water"
- WQ 15 "Bacterial Contamination of Household Water"
- WQ 16 "Land Application of Manure"
- WQ 17 "Agriculture's Effect on Environmental Quality: Key Management Issues"
- WQ 18 Agriculture's Impact on Environmental Quality Land Application of Municipal and Industrial Biosolids
- PPP-21 Pesticides and Container Management
- PPP-24 Pesticides and the Label
- PPP-25 Pesticides and Applicator Certification
- PPP-26 Pesticides and Their Proper Storage
- PPP-28 Pesticides and Spill Management
Water Quality Protection: Frank Answers to the 20 Most Frequently Asked Questions. The Alliance for a Clean Rural Environment (ACRE), Kansas City, MO.
Peterson, Doug, Ed Giles, Michael C. Hirschi and F. William Simmons. This Land: 50 Ways Farmers Can Protect Their Ground water. C1324, pp 78-88. University of Illinois, Urbana, College of Agriculture, Cooperative Extension Service, July 1993.
Rao, P.S.C. and A.G. Hornsby. Behavior Of Pesticides in Soil and Water. SL 40 (revised). Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida, Gainesville. October 1989.
Waldron, Acie C. Pesticides and Groundwater Contamination. Bulletin 820. Ohio Cooperative Extension Service, The Ohio State University. April 1992
Thanks to Kerry McCarthy, a 1993 Purdue College of Agriculture graduate, for her initial work on this publication.
Cooperative Extension work in Agriculture and Home Economics, state of Indiana, Purdue University, and U.S. Department of Agriculture cooperating; H.A. Wadsworth, Director, West Lafayette, IN. Issued in furtherance of the acts of May 8 and June 30, 1914. The Cooperative Extension Service of Purdue University is an affirmative action/equal opportunity institution.